Brand:-
Model:-
MOQ:-0 -
Antibiotic Drugs Tigecycline For Bacterial Infections CAS 220620-09-7
| Name | Tigecycline |
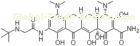
| Synonyms | (4S,4aS,5aR,12aS)-4,7-Bis(dimethylamino)-9-[(tert-butylamino)acetamido]-3,10,12,12a-tetrahydroxy-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracen-2-carboxamide |
| Molecular Formula | C29H39N5O8 |
| Molecular Weight | 585.65 |
| CAS Registry Number | 220620-09-7 |
Description:
Tigecycline is an antibiotic used to treat a number of bacterial infections.It is a first in class glycylcycline that is administered intravenously. It was developed in response to the growing rate of Antibiotic resistance in bacteria such as Staphylococcus aureus, Acinetobacter baumannii, and E. coli.As a tetracycline derivative antibiotic, its structural modifications has expanded its therapeutic activity to include Gram-positive and Gram-negative organisms, including those of multi-drug resistance. It is approved to treat complicated skin and soft tissue infections, complicated intra-abdominal infections, and community-acquired bacterial pneumonia in individuals 18 years and older.
COMPANY INTRODUCTION:
Our company, as one of the most experienced exporter in China,specializes in API
and medicine intermediates. We export large quantity to USA,Europe and many other
countries all of the world, deal to high quality and reasonable price.
Patent Disclaimer:
1. Products protected by valid patents are not offered for sale in jurisdictions where the sale of such products constitutes a patent infringement. The current list only reflects the products and technologies that are available, under development: note that some products may be developed or produced for internal and experimental uses with no commercal aim.
2. Sales of products are limited to those allowed by Chapter VII PLPRC 63, the above includes Research and development quantities.
3. R&D use in accordance with (1) 35 USC 271(e)+A13(1) in the U.S.; (2) Section 69.1 of Japanese Patent Law in Japan; (3) Section 11, No. 2 of the German Patent Act of 1981 in Germany; (iv) Section 60, Paragraph 5b of the U.K. Patents Act of 1977 in the U.K.; (4) Sections 55.2(1) and 55.2(6) and other common law exemptions of Canadian patent law; (5) Section 68B of the Patents Act of 1953 in New Zealand together with the amendment of same by the Statutes Amendment Bill of 2002; (6) such related legislation and/or case law as may be or become applicable in the aforementioned countries; and (7) such similar laws and rules as may apply in various other countries.